OHSIPP Position Statement
Our Society supports the use of indirect decompression systems, also known as interspinous spacers or interspinous process distraction systems without fusion for patients diagnosed with moderate stenosis. Patients afflicted with the condition do not have credible alternatives, albeit surgical or conservative[1]. Based upon the review of the body of peer-reviewed published evidence, approval by the United States Food and Drug Administration (FDA), real world experience and long-term patient outcomes, OHSIPP recommends qualified physicians consider use of these minimally invasive interventions and inform patients in their treatment plans based upon the clinical need and presentation. Because of proven safety and durable effectiveness, standalone indirect decompression systems are within the clinical community’s standard of care for their indicated use. At the time of this publishing, the Superion indirect decompression system is the only FDA approved Indirect Decompression System without fusion marketed in the US.
Based upon the body of Level I-IV peer-reviewed published evidence, FDA approval, and demonstration of cost-effectiveness, OHSIPP further recommends policymakers and payers enable timely access to indirect decompression systems when prescribed by a qualified physician who has used his or her best medical judgement for care most suitable to the individual diagnosed with moderate lumbar spinal stenosis.
Moderate Stenosis: Burden of Disease
Spinal stenosis refers to a a narrowing of the spnal column or spinal anatomy in the areas of the central canal, lateral recess, and/or neural foramina. Stenosis may be congenital, but more likely degenerative in origin. Lumbar spinal stenosis affects more than 200,000 people in the United States and is considered the most common reason for spinal surgery in patients >65 years[2].
In a claims-bases analysis, Parenteau et al (2021) reported the prevalence of a stenosis diagnosis over the age of 65 was >5% of the U.S. Medicare population, with women reporting a slightly higher prevalence than men[3]. Prevalence of lumbar spinal stenosis increases with age and body mass index[4].
While the degree of stenosis on the imaging study is important, in clinical practice the clinician should correlate the clinical findings with the degree of stenosis evident on the imaging study. As with interpretation of all imaging studies, clinical correlation is important. In the study that supported FDA approval and the indications for use, moderate degenerative lumbar spinal stenosis, is defined as follows:
25% to 50% reduction in the central canal and/or nerve root canal (subarticular, neuroforaminal) compared to the adjacent levels on radiographic studies, with radiographic confirmation of any one of the following:
Evidence of thecal sac and/or cauda equina compression
Evidence of nerve root impingement (displacement or compression) by either osseous or non-osseous elements
Evidence of hypertrophic facets with canal encroachment
AND associated with the following clinical signs:
Presents with moderately impaired Physical Function (PF) defined as a score of ≥ 2.0 of the Zurich Claudication Questionnaire (ZCQ)
Ability to sit for 50 minutes without pain and to walk 50 feet or more.
Clinical Presentation
Primary symptoms associated lumbar spinal stenosis include neurogenic claudication with back and leg pain, sensory loss, as well as weakness in the legs. More pronounced spinal stenosis may be presented through lower extremity weakness, muscle cramping, numbness and imbalance in gait. Symptoms are exacerbated by standing or walking, and extension of the spine. Short term relief may be observed when the patient is sitting or in flexion.
In addition to neurogenic claudication, lumbar spinal stenosis can present with symptoms that are more radicular in nature. As reported by Genevay and Atlas (2010)[5], “[u]nlike neurogenic claudication that is more commonly bilateral and associated with central canal stenosis, radicular symptoms due to spinal stenosis are more often unilateral and related to stenosis of the lateral recess or the foraminal canal. In these cases, patients tend to be younger and often have pain at rest and at night which is increased by the Valsalva maneuver. Leg pain is often described as severe and radicular in distribution and may be exacerbated with lumbar extension to the painful side (Kemp’s test)[6]. Examination findings may include a limited lumbar range of motion especially in extension, focal motor weakness in a specific root distribution, variable straight-leg tension signs, and diminished subjective sensation and reflexes in specific root distributions”. In adults over the age of sixty, spondylosis (degenerative arthritis affecting the spine) is the most common cause of stenosis.
In their expert consensus on identifying the top six factors most important in the clinical diagnosis of lumbar spinal stenosis, Tomkins-Lane et al (2016)[7] reported "leg or buttock pain while walking," "flex forward to relieve symptoms," "feel relief when using a shopping cart or bicycle," "motor or sensory disturbance while walking," "normal and symmetric foot pulses," "lower extremity weakness," and "low back pain."
Poor Operative and Non-Operative Alternatives
Conservative options including physiotherapy, bracing, cane, opioid and non-opioid medications, and exercises are offered, but in practice, the lack of consistent and durable relief with these options decreases the usefulness for the patients afflicted with spinal stenosis. Even epidural injections, with or without steroids, though effective in some cases, are often precluded due to the dose of steroids that the patient can receive[8]. In many cases, the epidural injections may provide temporary relief, but over the longer term, benefits of the therapy fade, leading the patients to seek surgical solutions. Cairns et al found persistent conservative care (>12 weeks) for lumbar spinal stenosis showed only minimal improvement in pain and function. Compared with extending conservative therapies or traditional spine surgery, interspinous lumbar decompression reduces both direct and indirect costs associated with lumbar spinal stenosis. Additionally, the costs of these conservative care options are not insignificant[9]. Nonetheless, contemporary algorithms advocate for conservative care before indirect decompression systems [10]. Diwan et al (2019) recommend the use of minimally invasive indirect decompression systems which deliver indirect decompression for moderate lumbar spinal stenosis after a treatment of 6 months of conservative care. OHSIPP recommends conservative care for at least 6 months, the choice of what options for which should be individualized to the specific needs of the patients and be at the discretion of the treating physician.
The other extreme of the therapeutic spectrum viz open spinal surgery - with or without fusion - is reserved for those with severe spinal stenosis, cauda equina syndrome, instability, or severe scoliosis. This is because the benefits of surgery, even in the best-case scenarios is time limited, the perioperative morbidity and mortality are higher with open spinal surgeries, and the hospital stays and post-surgical rehabilitation requiring skilled nursing facility costs are greater with open spine surgeries[11]. Published literature questions the benefits of complex fusion over simple laminectomy[12]. Regardless of outcomes, the rates of simple decompression surgery and simple fusions have declined, while complex fusion surgery increased from 1.3 per 100,000 (just under 1% of operations) to 19.9 per 100,000 (14.6% of operations), a 15-fold increase (2002-2007). Adjusted mean hospital charges for complex fusion procedures were $80,888 compared to $23,724 for decompression alone[13]. Thus, there is a large unmet need, and a void in the therapeutic armamentarium[14].
Even if surgery was a credible option, in many cases the concurrent co-morbidities preclude surgical intervention due to the perioperative morbidity and mortality. In such cases, indirect decompression systems may be the only credible and durable option[15].
Physician Qualification & Patient Selection
Implantation of indirect decompression systems without fusion should be performed only by qualified physicians, trained in the management of patients suffering from lumbar spinal stenosis and experienced in the placement of devices for whom patients would be indicated.
Conservative care of no less than six months in duration should be provided to patients before implantation of indirect decompression systems. Physicians must be granted latitude based upon their clinical training, experience and what the physician and patient determine are best for the individual based upon circumstances unique to the individual. Conservative care may include, but is not limited to:
Physical therapy for a duration of four to six weeks may be employed to reduce patient pain, disability and reliance of pain medication[16]. Studies report patients who have undergone physical therapy in advance of surgery furthermore enjoy faster recovery times[17].
Modification in the patient’s activities of daily living may be considered. Sustainability and patient compliance, however, must be considered for each patient relative to other clinical and psychological needs, age, occupation, vocation, and geography.
Oral medications including the use of non-narcotic analgesics and opiates may be prescribed. Physicians must use care and avoid negative drug-to-drug interactions and consider long-term effects of medications on other anatomical systems (e.g. longer-term use of NSAIDs and their association with gastrointestinal issues)[18]. Neuropathic medications are ineffective and often are poorly tolerated by the elderly affected with the condition, frequently leading to discontinuation[19]. Thus, while the CDC 2016 guidelines mandates the use of non-opioid therapies or allows opioid doses of up to 90mg morphine equivalent[20], these drugs are neither safe nor effective for the elderly patients afflicted with symptomatic neurogenic intermittent claudication pain spinal stenosis. Caution is warranted in the use of opioid therapy for the elderly, given the paucity of published evidence demonstrating efficacy for lumbar spinal stenosis in this population. Opiates are known to cause dependence, drowsiness, constipation (symptoms that are of particular concern in the elderly) and prove costly to the patient and health care system[21].
Epidural steroid injections may be used to relieve neck, arm, leg and back pain caused by inflamed spinal nerves from lumbar spinal stenosis. OHSIPP published guidelines recommend the use of fluoroscopically-guided caudal epidural injections, as well as for fluoroscopically guided lumbar interlaminar epidural injections. The evidence for lumbar transforaminal epidural injections is Level IV to III with moderate recommendation with fluoroscopically guided lumbar transforaminal epidural injections for long-term improvement. The evidence for percutaneous adhesiolysis in lumbar spinal stenosis is based on relevant, moderate to high quality randomized controlled clinical trials, observational studies, and systematic reviews[22].
Patients refractory to conservative care, or those for whom conservative therapies are not indicated and who otherwise meet the indications for use of indirect decompression systems without fusion for moderate stenosis should be considered. Considering poor outcomes with the conservative options, both independent reviews and Health and Human Services (HHS) best practices report call for early interventional pain management evaluation and treatment of all pain patients, including the elderly afflicted with spinal stenosis[23]. In the Joint OHSIPP-NANS 2019 fact sheet, OHSIPP reiterated its support of the 2019 HHS Best practices report that advocates for interventional pain therapies including indirect decompression systems.
Imaging studies are correlative with presentation of the patient’s signs and symptoms. Given their high sensitivity and specificity, magnetic resonance imaging (MRI) or computed tomography (CT) myelogram studies may be used to confirm diagnosis of lumbar spinal stenosis[24].
Treatment options, risks associated with conservative care, epidural injections, opioid use and surgical options must be well understood by the patient when developing care plans for the individual. Physicians must ensure informed consent of the patient.
The use of indirect decompression systems are contraindicated for patients with severe osteoporosis, spondylolisthesis with dynamic instability (as defined by >5mm movement between flexion and extension roentgenograms, greater than Grade 1 spondylolisthesis, allergies to titanium or titanium alloy, cauda equina syndrome, scoliosis (Cobb angle >10 degrees) and morbid obesity defined as a body mass index >40. Active systemic and local infection are also contraindications.
Likewise, patients with previous open laminectomy and or fusion at the target level are considered to be inappropriate, but not patients who have had minimally invasive lumbar spinal decompression or other percutaneous procedures where the spinous process and the lamina are preserved.
Evidence-Based Rationale
Indirect decompression systems used to treat moderate lumbar spinal stenosis are implanted posteriorly using minimally invasive techniques without disruption to the osseous or ligamentous tissue. Implantation typically occurs within the hospital outpatient or ambulatory surgical center, using cannulas under fluoroscopic guidance. Contraindications for indirect decompression systems include patients at risk for spinous process fracture (e.g. severe osteoporosis), spondylolisthesis with dynamic instability >than Grade 1[25]. Allergies to titanium or titanium alloy, cauda equina syndrome, scoliosis (Cobb angle >10 degrees) and morbid obesity defined as a body mass index >40.
Mechanisms of action associated with indirect decompression systems of the spinal cord and nerve roots lead to immediate symptom relief[26]. Cadaveric studies have shown increases in the spinal dimensions. For example, Falowski et al (2019)[27] Table 1 details increases in canal and foraminal dimensions following implantation of an indirect decompression system.
Results from a prospective, randomized controlled clinical trial were published by Patel et al (2015)[28]. This Level Ib evidence found the Superion indirect decompression system [Boston Scientific; Marlborough MA] relieved moderate lumbar spinal stenosis through two years post implant. Twenty-nine sites enrolled 391 patients, randomized to the index procedure or FDA approved control [X-STOP; Medtronic, Minneapolis MN]. At two years post implant, study subjects reported a 70% reduction in leg pain, 68% reduction in back pain, and clinical success measured by the Oswestry Disability Index (ODI) achieved in 65% of the patients. Superion success rates were reported as 99.5% for the index procedure, and 99.0% for the control.
There were no reported instances of device component fracture, disassembly or collapse. There was no device dislodgement for the index procedure, while 11.9% reported for control subjects. Use of the stand-alone indirect decompression system preserves treatment options and may obviate the need for decompressive laminectomy and or fusion in the majority of patients carefully selected and within the approved indications for use[29].
Long-term outcomes reported at five-years post implantation have demonstrated sustained and durable treatment effect. Nunley et al (2017)[30] reported 84% of patients demonstrated clinical success on at least two of three ZCQ domains. Individual ZCQ domain success rates were 75%, 81% and 90% for ZCQss, ZCQpf, and ZCQps, respectively. Leg and back pain success rates were 80% and 65%, respectively, and the success rate for ODI was 65%. Percentage improvements over baseline were 42%, 39%, 75%, 66%, and 58% for ZCQss, ZCQpf, leg and back pain VAS, and ODI, respectively (all P<0.001). Within-group effect sizes were classified as very large for four of five clinical outcomes (i.e., >1.0; all P<0.0001). Seventy-five percent of patients were free from reoperation, revision, or supplemental fixation at their index level at five years.
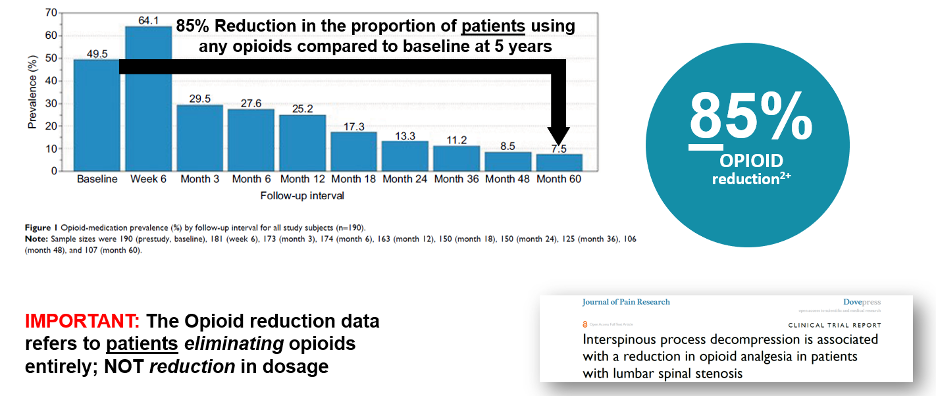
As noted earlier, physicians and patients must consider long-term effects of medications, drug interactions, as well as risks associated with opiates. This is particularly important when considering most patients are above the age of 65, may require polytherapy for treatment of other medical conditions or may be contraindicated for certain drugs or other therapeutic options. Standalone indirect decompression systems indicated for use in moderate stenosis patients have been shown to decrease the proportion of opioid users by 85% through five-years following the index procedure[31]. Specifically, a reduction in the proportion of those using opioids to manage their lumbar spinal stenosis symptoms.
To collect real-world outcomes, a registry for patients treated with interspinous indirect decompression spacers for lumbar spinal stenosis with intermittent neurogenic claudication was conducted. Tekmeyster et al (2019)[32] evaluated data from three-hundred sixteen physicians at 86 clinical sites located within the United States. Patient data were captured from in-person interviews and a phone survey. Outcomes included intraoperative blood loss, procedural time, leg and back pain severity (100 mm VAS), patient satisfaction and treatment approval at 3 weeks, 6 and 12 months. The mean age of registry patients was 73.0 ± 9.1 years of which 54% were female. Mean leg pain severity decreased from 76.6 ± 22.4 mm preoperatively to 30.4 ± 34.6 mm at 12 months, reflecting an overall 60% improvement. Corresponding responder rates were 64% (484 of 751), 72% (1,097 of 1,523) and 75% (317 of 423) at 3 weeks, 6 months and 12 months, respectively. Back pain severity improved from 76.8 ± 22.2 mm preoperatively to 39.9 ± 32.3 mm at 12 months (48% improvement); 12-month responder rate of 67% (297 of 441). For patient satisfaction at 3 weeks, 6 months and 12 months, 89%, 80%, and 80% were satisfied or somewhat satisfied with their treatment and 90%, 75%, and 75% would definitely or probably undergo the same treatment again. In the phone survey, the rate of revision was 3.6% (51 of 1,426).
For elderly patients suffering from significant comorbidities, implantation of indirect decompression systems were successfully shown to treat these patients at one or two levels. Doing so, Hartman et al (2019) reported avoidance of open spine surgery, anesthesia and risk of hospitalization commonly associated with this vulnerable patient population[33].
Diwan et al (2019) published their care algorithm based upon review of published evidence following inadequate response or failure of conservative care[34]. Researchers recommend the use of minimally invasive indirect decompression systems which deliver indirect decompression for moderate lumbar spinal stenosis. Implantation of these devices were supported due to long-term comparative trials and durability of treatment effect.
Cost Effectiveness
Using a Markov model evaluating cost-effectiveness of three treatment strategies for lumbar spinal stenosis, Parker et al (2015) concluded indirect decompression system implantation fell well below the QALY threshold of $50,000[35] and that such intervention versus sustained conservative care provided superior value. Cairns et al (2019) found persistent conservative care (>12 weeks) for lumbar spinal stenosis showed only minimal improvement in pain and function. Compared with extending conservative therapies or traditional spine surgery, indirect decompression system reduces both direct and indirect costs associated with lumbar spinal stenosis[36].
Conclusions & Recommendations
The body of Level I-IV published evidence, long-term outcomes demonstrating durable treatment effect, avoidance of more invasive procedures and drug therapies, as well as consideration for the patient populations most likely to be candidates for indirect decompression systems should be considered within the standards of care for moderate lumbar spinal stenosis.
Policymakers and payers are strongly encouraged to enable timely access to FDA approved or cleared technologies, when deemed medically necessary and indicated for this procedure.
References
[1] Diwan et al. An Algorithmic Approach to Treating Lumbar Spinal Stenosis: An Evidenced-Based Approach. Pain Med. 2019 Dec 1;20(Suppl 2):S23-S31. doi: 10.1093/pm/pnz133
[2] Lurie J, Tomkins-Lane C.. Management of lumbar spinal stenosis. BMJ 2016;352:h6234.
[3] Parenteau, C.S., Lau, E.C., Campbell, I.C. et al. Prevalence of spine degeneration diagnosis by type, age, gender, and obesity using Medicare data. Sci Rep 11, 5389 (2021).
[4] Kalichman, L., Guermazi, A., Li, L. & Hunter, D. J. Association between age, sex, BMI and CT-evaluated spinal degeneration features. J. Back Musculoskelet. Rehabil. 22, 189–195. (2009); Kalichman, L., Kim, D. H., Li, L., Guermazi, A. & Hunter, D. J. Computed tomography-evaluated features of spinal degeneration: prevalence, intercorrelation, and association with self-reported low back pain. Spine J. 10, 200–208. (2010).
[5] Genevay S, Atlas SJ. Lumbar spinal stenosis. Best Pract Res Clin Rheumatol. 2010 Apr;24(2):253-65. doi: 10.1016/j.berh.2009.11.001. PMID: 20227646; PMCID: PMC2841052.
[6] Jenis LG, An HS. Spine update. Lumbar foraminal stenosis. Spine 2000;25(3):389–94.
[7] Tomkins-Lane C, Melloh M, Lurie J, et al. ISSLS prize winner: Consensus on the clinical diagnosis of lumbar spinal stenosis: Results of an international Delphi study. Spine 2016;4115:1239–46.
[8] Manchikanti L, Knezevic NN, Navani A, Christo PJ, Limerick G, Calodney AK, Grider J, Harned ME, Cintron L, Gharibo CG, Shah S, Nampiaparampil DE, Candido KD, Soin A, Kaye AD, Kosanovic R, Magee TR, Beall DP, Atluri S, Gupta M, Helm Ii S, Wargo BW, Diwan S, Aydin SM, Boswell MV, Haney BW, Albers SL, Latchaw R, Abd-Elsayed A, Conn A, Hansen H, Simopoulos TT, Swicegood JR, Bryce DA, Singh V, Abdi S, Bakshi S, Buenaventura RM, Cabaret JA, Jameson J, Jha S, Kaye AM, Pasupuleti R, Rajput K, Sanapati MR, Sehgal N, Trescot AM, Racz GB, Gupta S, Sharma ML, Grami V, Parr AT, Knezevic E, Datta S, Patel KG, Tracy DH, Cordner HJ, Snook LT, Benyamin RM, Hirsch JA. Epidural Interventions in the Management of Chronic Spinal Pain: American Society of Interventional Pain Physicians (ASIPP) Comprehensive Evidence-Based Guidelines. Pain Physician. 2021 Jan;24(S1):S27-S208. PMID: 33492918.
[9] Cairns K et al. Cost-effectiveness and Safety of Interspinous Process Decompression (Superion)Pain Medicine, 20(S2), 2019, S2–S8 Review article
[10] Diwan et al. An Algorithmic Approach to Treating Lumbar Spinal Stenosis: An Evidenced-Based Approach. Pain Med. 2019 Dec 1;20(Suppl 2):S23-S31. doi: 10.1093/pm/pnz133).
[11] Ghogawala Z, et al.: Laminectomy plus Fusion versus Laminectomy Alone for Lumbar Spondylolisthesis. NEJM 2016; Weinstein JN, Tosteson TD, Lurie JD, et al. Surgical versus nonsurgical therapy for lumbar spinal stenosis (2 years) N Engl J Med. 2008;358(8):794-810; Weinstein JN, et al.: Surgical versus Non-Operative Treatment for Lumbar Spinal Stenosis. Four-Year Results of the Spine Patient Outcomes Research Trial (SPORT). Spine 2010.
[12] Forsth P et al. A Randomized, Controlled Trial of Fusion Surgery for Lumbar Spinal Stenosis. N Engl J Med 2016; 374:1413-1423).
[13] Deyo RA et al. Trends, Major Medical Complications, and Charges Associated with Surgery for Lumbar Spinal Stenosis in Older Adults. JAMA 2010; 303,1259-1265.
[14] Lauryssen C, et al.: Stand-alone interspinous spacer versus decompressive laminectomy for treatment of lumbar spinal stenosis. Expert Rev Med Devices 2015; 12(6):763-769.
[15] Hartman J, Granville M, Jacobson RE. The Use of Vertiflex® Interspinous Spacer Device in Patients With Lumbar Spinal Stenosis and Concurrent Medical Comorbidities. Cureus. 2019 Aug 12;11(8):e5374. doi: 10.7759/cureus.5374. PMID: 31616607; PMCID: PMC6786837.
[16] Slater J, Kolber MJ, Schellhase KC, et al. The influence of exercise on perceived pain and disability in patients with lumbar spinal stenosis. A systematic review of randomized controlled trials. Am J Lifestyle Med 2016;102:136–47.
[17] Lindback Y, Tropp H, Enthoven P, Abbott A, Oberg B.. PREPARE: Presurgery physiotherapy for patients with degenerative lumbar spine disorder: A randomized controlled trial. Spine J 2018;188:1347–55.
[18] Kuritzky L, Samraj GP. Nonsteroidal anti-inflammatory drugs in the treatment of low back pain. J Pain Res. 2012;5:579-590. doi:10.2147/JPR.S6775.
[19] Finnerup NB, et al. Pharmacotherapy for neuropathic pain in adults: A systematic review and meta-analysis. Lancet Neurol 2015; 14:162–73.
[20] Dowell, D., Haegerich, T. M., & Chou R: CDC Guideline for Prescribing Opioids for Chronic Pain-United States, 2016 Morbidity and Mortality Weekly Report. JAMA - J Am Med Assoc 2016 at <http://www.cdc.gov/mmwr/cme/conted.html.
[21] Adogwa O, Davison MA, Vuong VD, et al.. Long term costs of maximum non-operative treatments in patients with symptomatic lumbar stenosis or spondylolisthesis that ultimately required surgery: A five-year cost analysis.
[22] Manchikanti L, Knezevic NN, Navani A, Christo PJ, Limerick G, Calodney AK, Grider J, Harned ME, Cintron L, Gharibo CG, Shah S, Nampiaparampil DE, Candido KD, Soin A, Kaye AD, Kosanovic R, Magee TR, Beall DP, Atluri S, Gupta M, Helm Ii S, Wargo BW, Diwan S, Aydin SM, Boswell MV, Haney BW, Albers SL, Latchaw R, Abd-Elsayed A, Conn A, Hansen H, Simopoulos TT, Swicegood JR, Bryce DA, Singh V, Abdi S, Bakshi S, Buenaventura RM, Cabaret JA, Jameson J, Jha S, Kaye AM, Pasupuleti R, Rajput K, Sanapati MR, Sehgal N, Trescot AM, Racz GB, Gupta S, Sharma ML, Grami V, Parr AT, Knezevic E, Datta S, Patel KG, Tracy DH, Cordner HJ, Snook LT, Benyamin RM, Hirsch JA. Epidural Interventions in the Management of Chronic Spinal Pain: American Society of Interventional Pain Physicians (ASIPP) Comprehensive Evidence-Based Guidelines. Pain Physician. 2021 Jan;24(S1):S27-S208. PMID: 33492918.
[23] Bates D et al. A Comprehensive Algorithm for Management of Neuropathic Pain. Pain Medicine, 20, 2019, S2–S12; U.S. Department of Health and Human Services (2019, May). Pain Management Best Practices Inter-Agency Task Force Report: Updates, Gaps, Inconsistencies, and Recommendations. Retrieved from U. S. Department of Health and Human Services website: https://www.hhs.gov/ash/advisory-committees/pain/reports/index.html. ASIPP-NANS factsheet).
[24] de Schepper EI, Overdevest GM, Suri P, et al. Diagnosis of lumbar spinal stenosis: An updated systematic review of the accuracy of diagnostic tests. Spine 2013;388:E469–81.
[25] Reference: Superion Interspinous Spacer, FDA PMA approval order and indications for use (P140004) accessed at: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfPMA/pma.cfm?id=P140004.
[26] Block JE, Lavelle WF, Nunley PD. Toward a cure for lumbar spinal stenosis: The potential of interspinous process decompression. Med Hypotheses. 2019 Nov;132:109357. doi: 10.1016/j.mehy.2019.109357. Epub 2019 Aug 10. PMID: 31421414.
[27] Falowski SM, Sayed D, Deer TR, Brescacin D, Liang K. Biomechanics and Mechanism of Action of Indirect Lumbar Decompression and the Evolution of a Stand-alone Spinous Process Spacer. Pain Med. 2019 Dec 1;20(Suppl 2):S14-S22. doi: 10.1093/pm/pnz129. PMID: 31808533; PMCID: PMC7101165.
[28] Patel VV, Whang PG, Haley TR, Bradley WD, Nunley PD, Davis RP, Miller LE, Block JE, Geisler FH. Superion interspinous process spacer for intermittent neurogenic claudication secondary to moderate lumbar spinal stenosis: two-year results from a randomized controlled FDA-IDE pivotal trial. Spine (Phila Pa 1976). 2015 Mar 1;40(5):275-82. doi: 10.1097/BRS.0000000000000735. PMID: 25494323.
[29] Lauryssen C, Jackson RJ, Baron JM, Tallarico RA, Lavelle WF, Deutsch H, Block JE, Geisler FH. Stand-alone interspinous spacer versus decompressive laminectomy for treatment of lumbar spinal stenosis. Expert Rev Med Devices. 2015;12(6):763-9. doi: 10.1586/17434440.2015.1100071. Epub 2015 Oct 21. PMID: 26487285.
[30] Nunley PD, Patel VV, Orndorff DG, Lavelle WF, Block JE, Geisler FH. Five-year durability of stand-alone interspinous process decompression for lumbar spinal stenosis. Clin Interv Aging. 2017 Sep 6;12:1409-1417. doi: 10.2147/CIA.S143503. PMID: 28919727; PMCID: PMC5593396.
[31] Nunley PD, Deer TR, Benyamin RM, Staats PS, Block JE. Interspinous process decompression is associated with a reduction in opioid analgesia in patients with lumbar spinal stenosis. J Pain Res. 2018 Nov 20;11:2943-2948. doi: 10.2147/JPR.S182322. PMID: 30538533; PMCID: PMC6251434.
[32] Tekmyster G, Sayed D, Cairns KD, Raso LJ, Kim C, Block JE. Interspinous Process Decompression With The Superion Spacer For Lumbar Spinal Stenosis: Real-World Experience From A Device Registry. Med Devices (Auckl). 2019 Oct 3;12:423-427. doi: 10.2147/MDER.S220431. PMID: 31632160; PMCID: PMC6781846.
[33] Hartman J, Granville M, Jacobson RE. The Use of Vertiflex® Interspinous Spacer Device in Patients With Lumbar Spinal Stenosis and Concurrent Medical Comorbidities. Cureus. 2019 Aug 12;11(8):e5374. doi: 10.7759/cureus.5374. PMID: 31616607; PMCID: PMC6786837.
[34] Diwan S, Sayed D, Deer TR, Salomons A, Liang K. An Algorithmic Approach to Treating Lumbar Spinal Stenosis: An Evidenced-Based Approach. Pain Med. 2019;20(Suppl 2):S23-S31. doi:10.1093/pm/pnz133.
[35] Parker SL, Anderson LH, Nelson T, Patel VV. Cost-effectiveness of three treatment strategies for lumbar spinal stenosis: Conservative care, laminectomy, and the Superion interspinous spacer. Int J Spine Surg. 2015 Jul 9;9:28. doi: 10.14444/2028. PMID: 26273546; PMCID: PMC4528571.
[36] Cairns K, Deer T, Sayed D, van Noort K, Liang K. Cost-effectiveness and Safety of Interspinous Process Decompression (Superion). Pain Med. 2019 Dec 1;20(Suppl 2):S2-S8. doi: 10.1093/pm/pnz245. PMID: 31808529; PMCID: PMC6896024.